
I introduced exergy to readers 4.5 years ago, had a refresher on it for industrial decarbonization earlier this year, and it is part of our decarbonization training course that is frontrunning AESP’s annual conference in Nashville on February 7th.
Preserving v Wasting Energy
Ok. Exergy is the maximum available energy in a resource until it reaches thermal equilibrium with its surroundings.
Example 1: You’re on top of a hill with your bicycle, and you want to coast as far as you can without peddling or cheating with one of those lazy electric bikes. What do you do? You crouch down into a dangerous riding position like a cycling professional (NOT recommended) to go as fast and as far as you can – or ride like this guy (NOT recommended).

Image Source: Bicycling.com
Example 2: You take a 100°F shower for 15 minutes with an electric water heater powered by a thermal power plant that’s rejecting heat through iconic natural convection cooling towers. The heated shower water leaves the house losing its heat to the sanitary sewer and soil.
Example 1 wastes almost zero exergy because the only heat lost to the surroundings is a little bearing friction and airflow drag (friction). Example 2 is horrible. The power plant is dumping gobs of heat that isn’t close to the temperature of the surroundings, and at home, more heat is lost to the ground. You may have seen that only some puny percentage of energy we consume, like 3%, is beneficial. There it is.
Homely But Smart
All the sex appeal with decarbonization belongs with electric vehicles, solar panels, uber complex control networks, and wildly expensive batteries with varied crazy ways of storing electricity. Overlooked are far more cost-effective, concentrated sources of waste to be harvested before rejection to the surroundings.
How homely are these solutions? The inspiration came from a magazine called Today’s Boiler. Yep. I still get some printed materials in my mailbox, and frankly, print gets my attention over thousands of emails per week.
The article states decarbonization requires massive amounts of renewable generation and people are getting annoyed with wind farms and solar fields. Solar fields are ugly, whether they cover what used to be a pasture or an apple orchard. Instead, the author says, let’s stop throwing energy away. I agree. He writes, “Waste heat is the energy contained in a fluid or a product that is usually left to dissipate in the atmosphere [lost exergy] or through cooling fluids, either because the quantity is too small, the temperature too low, or the heat transfer requirement to recover that energy is too complex. That recovered energy can be used for either electricity generation or to improve the process energy efficiency. Numerous examples abound. In reality, even today, a large portion of the industrial energy input is lost as waste heat.”
Heat Transfer
Energy-intensive manufacturing can be divided into two classes. The first class needs heating and cooling at the same time. Most food processing falls into this category. They need lots of hot water or steam for cleaning, pasteurizing, peeling potatoes, or puffing cereal and salty snacks with steam. They need refrigeration or dehumidification to preserve food and keep lines running without caking and clogging. They burn energy to make heat, and at the same time, they burn energy to refrigerate or dehumidify/cool the manufacturing spaces. This is a huge lost opportunity because heat can be moved from the refrigeration/cooling loads to the heating loads, maybe with half the energy to produce only one of them.
The second class includes manufacturers that require tremendous amounts of heat. These include glass, primary metals (e.g., steel), and foundries. Waste streams of heat from these processes are so hot that exhaust plenums are water-cooled heat exchangers to keep them from melting. These facilities don’t need cooling below temperatures that air provides, which is to say, they don’t need refrigeration. The opportunities with these processes include preheating feedstock with waste heat streams OR generating electricity with waste heat.
Power Cycle Efficiency
From my first post on exergy, the efficiency of any engine or power cycle, including thermal power generation, is mainly a function of the high temperature. From my first week in thermodynamics (Carnot pronounced car-no):
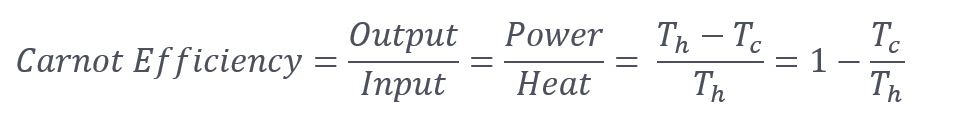
Where:
T = absolute temperature
And h and c subscripts are hot and cold, respectively.
We are mostly stuck with Tc, which is typically surrounding air temperature in this case. You can see we want a high Th for good efficiency. Waste heat at 500°F (Th) or more can produce electricity at decent efficiency, say 15%, considering the heat is free. To increase efficiency, Th can be boosted by adding some heat to the cycle with a very high incremental efficiency.
Returns
The challenge with these massive energy savers is “low” return on investment, which might be three to eight years’ simple payback. This isn’t good enough for manufacturers, which typically want simple paybacks of 1-2 years. What’s the ROI for solar panels and wind turbines? Utility weighted cost of capital is typically 10-15%. That’s not as good as the waste heat resources. What are we talking about, resource-wise? Two megawatts of electricity from one sheet-glass factory. That’s a wind turbine, except it runs all the time.
But more importantly, waste heat recovery coincides almost perfectly with energy demand, unlike renewable supplies, which are only available when the sun shines, and the wind blows. Reliable power when you need it is much more valuable than power that comes and goes. This is where we are – matching demand to supply.
Investing
Energy from waste streams is like renewable energy, but better. It’s free energy for the taking, so where are the production tax credits and investors? Let’s open it up!


