
Last week in Thermal Storage for Grid-Interactive Efficient Buildings (GEBs), I introduced the importance of phase changes from solid to liquid to vapor, and the reverse, to our modern world. Benefits include heating, cooling, and refrigeration for all types of uses, including space conditioning, food storage and transportation, healthcare, manufacturing, and of course, thermal storage.
The simplest everyday thermal storage material is ice. Grocery and convenience companies have enormous ice-making facilities for guess what: thermal storage and your lowly Igloo or swanky Yeti coolers. I have a Coleman Lil Oscar cooler that I’ve used since Ha School. Now that is sustainability! Yes, it most certainly has lots of miles and usage.
 Applications
Applications
There are ways to use thermal mass of buildings and their contents for thermal storage. Massive, as in brick or cut stone, buildings require less heating and cooling energy and lower peak demand because of their inherent thermal storage characteristics.
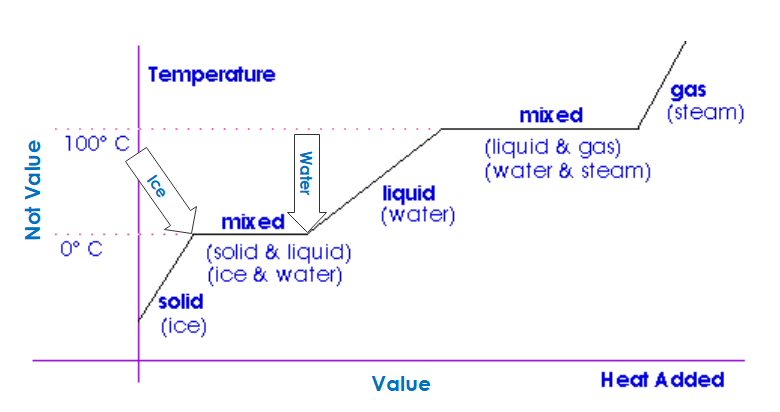
Beyond that, we can pre-cool or pre-heat the building’s mass, including partitions (interior walls), furniture, floors, food, and beverages in storage and retail facilities. However, this is quite limited per the chart last week, showing that temperature change (vertical axis) is undesirable. People and products will accommodate some temperature change, but it is very limited.
 Minimal Opportunities
Minimal Opportunities
For example, I would not advise using thermal storage via temperature change in a retail store’s produce section. Sub-cooling products in the dairy and meat sections may be worth considering. There are only a few degrees available to avoid the onset of freezing, but there is a lot of mass (water) in these products for thermal storage. Non-perishable goods like beer, soda, water, and sports drinks offer the most refrigerated storage capacity because they won’t be damaged or “go bad” if they drift a few degrees above setpoint.
Limited Opportunities
Frozen food storage is the best opportunity because, as far as I know, nothing will be substantially damaged by driving it’s temperature substantially below setpoint to coast through an on-peak period. Subcooling ice cream from -5F to -25F and letting it coast back to -5F during the on-peak period will not damage the product.
Interestingly, food storage gurus say ice cream should be stored at -20F, even in supermarkets. This jibes with an old guy I worked with in the industry years ago. But yet, if you check out your favorite supermarket freezer case, I’ll bet it’s closer to 0F or -5F at most. Cheaters.
Greatest Opportunities
The greatest opportunities for thermal storage involves phase change. Food products are not viable candidates for freeze, thaw, freeze, thaw. And while people often complain that they are freezing (you know these people), they are “freezing” at roughly 97F.
So let’s get into it!
Ice Ice Baby
Ice is the most common, old school means of storing thermal energy. Check out these old photos from industrial-scale ice harvesting from back in the day. In many ways, our ancestors lived a much more sustainable lifestyle than we do today.
CALMAC is a product that’s been around for decades. It circulates brine from a chiller through a heat exchanger in giant garbage cans to freeze water during off-peak hours for use in on-peak hours. An X-RAY depiction of one of these units is shown below. A photo of a farm of storage tanks follows. They’re big.
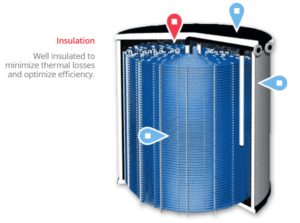 Ice is used for thermal storage to cool buildings because of the phase change that I’ve described many times. A lot of cooling capacity can be stored in a small space compared to storing chilled water.
Ice is used for thermal storage to cool buildings because of the phase change that I’ve described many times. A lot of cooling capacity can be stored in a small space compared to storing chilled water.
However, ice isn’t the best from an efficiency standpoint because it is far away from the temperature of the load being served, which is 55F supply air to maintain an office space or some other occupied space at 74F.
 Water freezes at 32, and therefore, to make ice, antifreeze has to be added to the chilled water loop to keep it from freezing. There are two or maybe three penalties with this. First, the chiller’s work to transfer heat from 25F for freezing the tanks to 85F condenser water outdoors requires extra energy compared to producing chilled water at 35F-45F, directly sent to space cooling. Refer to the cartoon below to follow along. You can see the system moves heat from 74F conditioned space temperature or 32F ice to whatever the hot outdoor conditions are at the time.
Water freezes at 32, and therefore, to make ice, antifreeze has to be added to the chilled water loop to keep it from freezing. There are two or maybe three penalties with this. First, the chiller’s work to transfer heat from 25F for freezing the tanks to 85F condenser water outdoors requires extra energy compared to producing chilled water at 35F-45F, directly sent to space cooling. Refer to the cartoon below to follow along. You can see the system moves heat from 74F conditioned space temperature or 32F ice to whatever the hot outdoor conditions are at the time.
The other penalty for this form of ice storage is the antifreeze. Antifreeze, while necessary, is more viscous, and therefore, takes more energy to pump. It also reduces heat transfer for a given temperature difference, all else equal.
 Not Ice
Not Ice
Making ice per the above is a relatively complicated mess. The cartoon shown is a demonstration of only what the chiller sees. Although ice storage systems have been engineered to supreme simplicity and scalability, they still require a lot of piping and extra pumping.
Rather than using an ice farm, imagine putting phase-change material in the conditioned space to store thermal energy. Now we’re talking! The benefits include:
- No messing with the existing cooling system, whether it’s for space conditioning or storing frozen food.
- Small changes in cooling system temperature setpoints, and therefore, minor efficiency/energy loss.
- Practically invisible and non-invasive.
We have analyzed one such phase change material for cold storage already. The system uses materials that look suspiciously like the ice packs I put in my Lil Oscar. See below. These storage units passively sit on top of storage racks, out of sight, out of mind, except when the electric bill comes.
 Some phase change materials can be easily modified to accommodate a finite range of storage temperatures – the temperatures at which they freeze and melt.
Some phase change materials can be easily modified to accommodate a finite range of storage temperatures – the temperatures at which they freeze and melt.
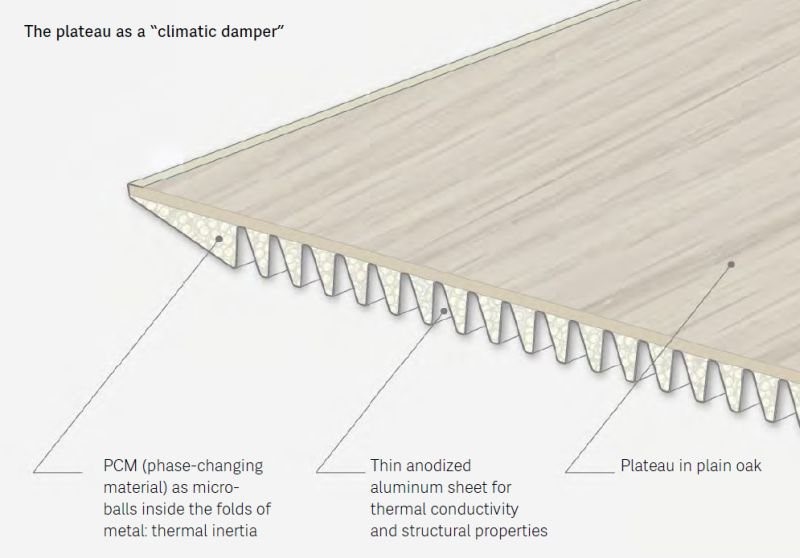
Why stop there! Phase change materials can be developed for anything in a building – tables, flooring, walls, and more. No one will know it’s there. Below is a diagram for such a system in the form of a tabletop.
I would say these things are coming – big time. My guess is they will bury batteries in the cost-effectiveness of energy storage.

 Minimal Opportunities
Minimal Opportunities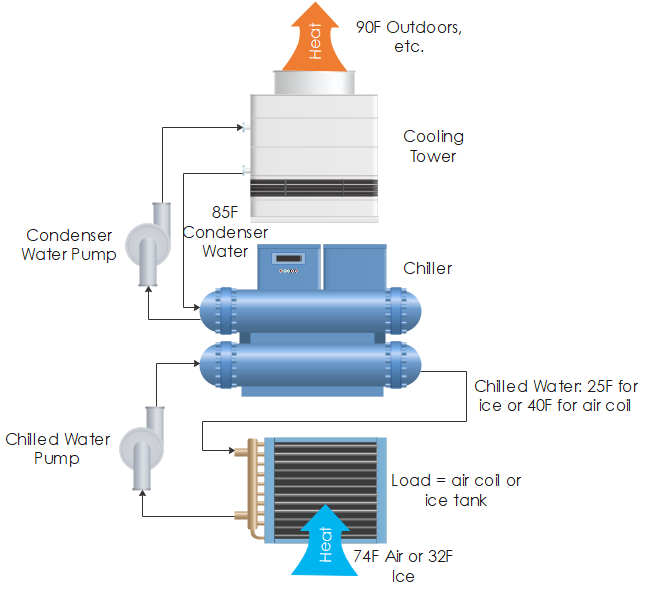 Not Ice
Not Ice