
Two weeks ago, in Natural Gas Savings – Traps and H Vacs we looked at some basic natural-gas-saving measures. The post was wildly popular, and I had a good time, so let’s do it again!
We compared some aspects of natural gas end-uses and waste that compare to compressed air. For example, we looked at steam traps versus compressed air leaks. Similarly, there are a lot of things that can be done with steam supply sources v compressed air sources, both including controls and other things.
Minerals
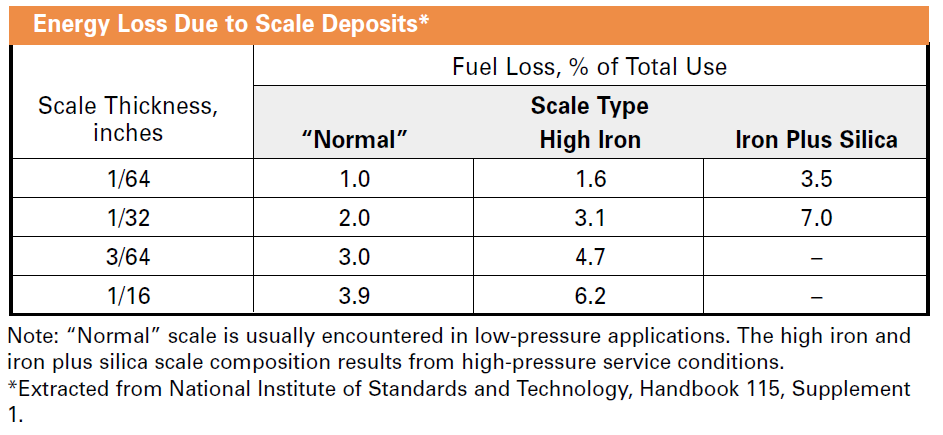
Tap water typically has a lot of dissolved minerals, including calcium, magnesium, and silica. These minerals are essential for good health, but they are bad for boiler efficiency. Since I just mentioned health, these minerals will plate out[1] on hot surfaces such as the waterside of the boiler heat exchanger – just as plaque plates out on artery walls and causes them to clog. Boiler plating (or scale) occurs as boiling on a microscopic level called nucleate boiling occurs. Nucleate boiling is that rumbling sound your pot of water makes before visual boiling takes place. Tiny steam bubbles form and collapse – rumbling. The water boils away leaving minerals, aka lime or scale, coating the heat transfer surface. Scale is a great insulator – not something we want for optimal boiler efficiency.
Tap water always leaves this white residue when water evaporates. It’s the film on glasses out of the dishwasher or the white powder left behind when tap water is used for adiabatic (fine mist) humidification.
Water Tubes and Fire Tubes
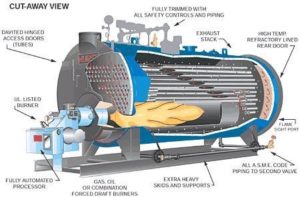
There are two types of boilers: water tube and fire tube. Water-tube boilers are used in large industrial applications, including power plants. Since water is inside the tubes, these boilers can produce higher pressure steam. A simplified cartoon[2] below shows how it works, but that, of course, is not what it looks like. For that, refer to the picture[3] next door.

Fire tube boilers are far more common for space heating and for industrial needs where high pressures are not required. The cartoon[4] below nicely depicts how it works and what it looks like for real. Combustion gasses are on the inside of the tubes, and the water is outside the tubes.
Makeup Water
Boilers continuously lose water to leaks – steam or water leaks, and in some cases, processes need direct steam. In both cases, water that leaves the boiler in gaseous form does not return.
Do you know how mass quantities of potatoes are peeled? They are not peeled by elves. They are peeled with steam. Steam is injected into a pressurized vat of potatoes to bring their surface temperature above the boiling point. Then the pressure is released, and the skin pops off as the water in and near the skin flashes into steam at atmospheric pressure. Here is one that peels 65 metric tons per hour. Dazzle your kids and pets with that one!
As pure water (steam) leaves the boiler, minerals would concentrate/accumulate in the boiler, eventually plating on heat exchanger surfaces to decrease heat transfer and efficiency. The traditional means to mitigate this is boiler blowdown, which dumps the concentrated mixture of minerals and water down the drain.
Waste Reduction
There are at least two ways to save energy when dealing with dissolved minerals in makeup (tap) water in boilers. The two we will examine are blowdown heat recovery and reverse osmosis.
Blowdown Heat Recovery
Blowdown heat recovery is pretty simple. Water is leaving the system due to leaks or process steam, so it must be replaced with tap water. The opportunity is to recover heat from the discharge of the blowdown rather than sending it down the drain at 210 degrees[5]. Heat recovery = natural gas savings.
Reverse Osmosis
Reverse osmosis is a sexy term for filtering. Filters catch chunks like coffee grounds so they don’t contaminate the product – a cup of coffee. Customers wouldn’t take kindly to chewing on the dregs of their last few sips of their beloved au natural brain chemical[6]. However, the filter for coffee is what I affectionately call a cat catcher. It is crude enough to stop large chunks like a fiberglass air filter prevents cats from circulating through your home furnace.
In the case of dissolved minerals in tap water, massively higher pressure is required to push water molecules through the filter, also known as a membrane, which filters the dissolved solids. This is called reverse osmosis. Osmosis is the diffusion of minerals to equalize across a membrane. Reverse osmosis is, therefore, the concentration of minerals (as in filter) on one side with purer dissolvent (water) on the other side.


Reverse osmosis removes the minerals (99.9% per this source), almost eliminating the need for blowdown. The cost for an “RO” system is not insignificant, and a portion of the natural gas savings is offset by the added electricity cost to run the RO system. Squishing the tap water through the membrane requires high pressure with a positive displacement pump (cylinders and pistons). Reverse osmosis systems significantly reduce the need for blowdowns and associated heat loss and keeps boilers tubes squeaky clean for optimal heat transfer and efficiency. Here is a cool video for how it works – what’s in those tubes? This one is used for water treatment plants.
[1] My term for sticking to surfaces. Others call it precipitating.
[2] https://thermodyneboilersblog.wordpress.com/2017/01/18/advantages-and-working-principle-of-water-tube-boiler/
[3] http://vijayengineeringworks.co.in/water_tube_bolier.php
[4] https://feedwater.co.uk/industrial-boiler-types/
[5] Water sent to sanitary sewer systems typically needs to be below a certain temperature and to accomplish this, it is mixed with tap water – another waste of energy and resources.
[6] Caffeine.


